Study of Photoactive Materials Used in Photo Electrochemical Cell for Solar Energy Conversion and Storage

DOI:
https://doi.org/10.54060/jase.v3i1.40Keywords:
Photo potential, Photocurrent, Fill factor, Conversion efficiency, Storage capacity of cellAbstract
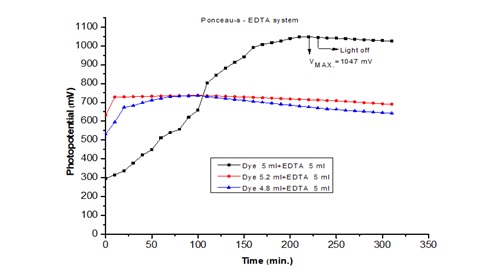
Photoelectrochemical Cell is a device that absorbs light with a high-absorption electrolyte solution and provides energy for photo chemical reactions. Ponceau-S was used as a photosensitizer and EDTA served as a reducing agent in the study of photoelectronchemical cells. The photocurrent and photo potential were 1047.0 mV and 390.0 µA respectively. The highest power of the cell was 84.0 µW, with a conversion efficiency of 1.61%. The fill factor of the cell was 0.20. The photoelectric cell can function at this power level for 240.0 minutes in storage (performance). The effects of various parameters on the cell's electrical output were observed. In this study, a mechanism for photocurrent generation in Photoelectrochemical cells is proposed.
Downloads
References
E. K. Rideal & D. C. Williams, “The action on the ferrous iodine iodide equilibrium,” J. Chem Soc, vol.127, pp.258-269, 1925.
E. Rabinowitch & L. F. Epstein, “Polymerization of Dyestuffs in Solution Thionine and Methylene Blue,” J. Chem. Phy, vol.63, no.1, pp.69-78, 1941.
E. Rabinowitch, L. F. Epstein, F. Karush, “A spectrophotometric study of thionine,” JOSA, vol.31, no.1, pp.77-84, 1941.
R. Gomar, “Photogalvanic cells,” Electochim Acta, vol.20, no.1, pp.13-20, 1975.
J. G. Calvert, A. M. Zarem, D. D. Erway, “Photochemical process for utilization of solar energy,” McGraw Hill New York, 1964.
W. D. K. Clark & J. A. Eckert, “Photogalvanic cells,” Sol. Energy, vol.17, no.3, pp.147-150, 1975.
M. Kaneko & A. Yamada, “Photopotential and photocurrent induced by a tolusafranine ethylenediaminetetraacetic acid system," The Journal of Physical Chemistry, vol.81, no.12, pp. 1213-1215, 1977.
A. S. N. Murthy, K. S. Reddy, “Studies on photogalvanic effect in systems containing toluidine blue,” Solar Energy, vol.30, no.1, pp.39-43, 1983.
M. Z. Hoffman, N. N. Lichtin, R. R. Hantala, et al., “Use of reductant and photosensitizer in photogalvanic cells for solar energy conversion and storage: oxalic acid–methylene blue system,” Solar Energy, N.N.Publisher, New Jersey,vol.141, no.2-3, pp. 175-177, 2001.
A. S. N. Murthy, H. C. Dark, K. S. Reddy, “Photogalvanic effect in riboflavin—ethylenediaminetetraacetic acid sys-tem." International Journal of Energy Research, vol.4, no.4, pp.339-343, 1980.
S. C. Amcta, S. Khamcsra, S. Lodha, et al., “Studies on photogalvanic effect in systems containing toluidine blue,” J. Photochem Photobiol, vol.81, 1989.
S. C. Ameta, S. Khamesra, M. Bala, et al., Use of Micelles in Photogalvanic Cell for Solar Energy Conversion and Stor-age,” The Philippine Journal of Science, vol.119, pp.371-373, 1990.
M. Mukhopadhyaya & B. B. Bhowmik, “Kinetics of photoinduced electron transfer in a photoelectrochemical cell con-sisting of thiazine dyes and Triton X-100 surfactant,” J. Photochem. Photobiol, vol.69, no.2, pp.223-227, 1992.
K. M. Gangotri, O. P. Regar, C. Lal, et al., “Role of an Anionic Surfactant and Reductant in Photo Galvanic Cell for So-lar Energy Conversion and Storage,” Int. J. Energy Res., vol.20, pp.581-585, 1996.
K. M. Gangotri, C. Lal, “Studies in Photogalvanic Effect and Mixed Dyes System: EDTA-Methylene Blue Toluidine Blue System,” International Journal of Energy Research, vol.24, pp.365-371, 2000.
K. M. Gangotri, C. Lal, “Use of Mixed Dyes in Photogalvanic Cells for Solar Energy Conversion and Storage: EDTA-Methylene Blue and Azur-B System,” Energy Sources, vol.23, no.3, pp.267-273, 2001.
K. M. Gangotri, R. C. Meena, “Use of reductant and photosensitizer in photogalvanic cells for solar energy conversion and storage: oxalic acid–methylene blue system,” J. Photochem Photobiol., vol.141, no.2-3, pp.175-177, 2001.
K. M. Gangotri, R. C. Meena, “Role of reductant and photosensitizer in solar energy conversion and storage: ascorbic acid-eiosin system,” J. Ind. Chem. Soc., vol.81, pp.576-578, 2004.
K. M. Gangotri & C. Lal, “Use of mixed dyes in photogalvanic cells for solar energy conversion and storage: EDTA-methylene blue and thionine system,” J. Power and Energy, vol.219, no.5, pp. 315-320, 2005.
R. C. Meena & R. S. Sindal, “USE OF SURFACTANT IN PHOTOGALVANIC CELL FOR. SOLAR ENERGY CONVERSION AND STORAGE: TWEEN-80-OXALIC ACID-TOLUIDINE BLUE SYSTEM,” Int. J.Chem. Sci., vol.2, no.3, pp.321-330, 2004.
R. C. Meena & G. Singh, “Role of reductant and photosensitizers in solar energy conversion and storage: Oxalic acid - Brilliant Cresyl system,” Int.J. Chem. Sci., vol.31, pp.463-470, 2006.
A. Chauhan, M. Genwa, I. Prakash, et al., “Study of photogalvanic effect in photogalvanic cell containing Azur B-NaLS-Ascorbic acid system,” J.Ind. Chem. Soc., vol.83, no.8, pp.799-802, 2006.
C. Lal, “Use of mixed dyes in a photogalvanic cell for solar energy conversion and storage: EDTA–thionine–azur-B sys-tem,” J. Power Sources, vol.164, no.2, pp.926-930, 2007.
K. R. Genwa & M. Genwa, “Photogalvanic cell: A new approach for green and sustainable chemistry,” Original Re-search Article Solar Energy Materials and Solar Cells, vol.92, no.5, pp.522-529, 2008.
K. R. Gemwa, A. Kumar, A. Sonel, “The Role of Ascorbic Acid in a Photogalvanic Solar Cell Containing a Crystal Vio-let-diocyle Sulphosuccinate System and to Study the Energy Efficiency of the Cell,” Original Research Article Applied Energy, pp.1431-1436, 2009.
K. M. Gangotri, V. Indora, “Studies in the photogalvanic effect in mixed reductants system for solar energy conversion and storage: Dextrose and Ethylenediaminetetraacetic acid–Azur A system,” Original Research Article Solar Energy, vol.84, no.2, pp.271-276, 2010.
M. Chandra, R. C. Meena, “Role of Ponceau-S-Mannitol System for Generation of Electrical Energy in Photogalvanic Cell,” Int. J. Chemical Science, vol.26, pp.1447-1456, 2010.
M. Chandra, R. C. Meena, “Role of reductant for generation of solar energy in photogalvanic cell,” J. Chem. And Pharm. Research, pp.264-270, 2011.
M. Chandra, A. Singh, R. C. Meena, “Role of Rose Bengal-Mannitol system for generation of electrical energy in pho-togalvanic cell,” Int. Chem. Sci., vol.7, no.42, pp.5642-5647, 2012.
R. C. Meena, Neelakshi, Manju, “Photochemical Study for Solar Energy Conversion & Storage in Solar Cell: Yellow 5GN-EDTA System,” Int. J. Chem. Pharmaceu. Sci., vol.18, pp.612-616, 2014.
N. Kumar, P. K. Meena, S. L. Meena, “Use of Photosensitizer in a Photoelectrochemical Cell for Solar Energy Conver-sion and Storage,” Journal of applicable chemistry, vol.7, no.6, pp.1682-1690, 2018.
K. Meena, S. R. Saini and R. C. Meena, “Studies of Surfactant in Photogalvanic Cell for Solar Energy Conversion and Storage,” Chemical Science international journal, vol.18, no.2, 2017.
S. R. Saini, S. L. Meena and R. C. Meena, “Studies of Surfactant in Photogalvanic Cell for Solar Energy Conversion and Storage,” Advance in Chemical Engineering and Science, vol.7, pp.125-136, 2017.
S. L. Meena, P. K. Meena, R. C. Meena, et al., “USE OF ACID RED-36 DYE AS PHOTOSENSITIZER IN PHOTOGALVANIC CELL FOR SOLAR ENERGY CONVERSION AND STORAGE,” Journal of Advance Scientific Research, vol.12, no.2, pp.110-116
Downloads
Published
How to Cite
CITATION COUNT
Issue
Section
License
Copyright (c) 2023 Shanker Lal Meena, Ravi Kumar Bhupesh, Lal Chand Yadav

This work is licensed under a Creative Commons Attribution 4.0 International License.



























